I tend to go through finishes, and especially paint fairly slowly, so I lose a lot to the finish skinning over in the can, and then have to either filter (losing more material) or be really careful to avoid the leftover bits and blobs. This wasn't so bad back when a gallon of good paint was $15, but now it's pushing $80, and I'm a cheapskate. Hence Bloxygen. But being a cheapskate it was hard to swallow the price of bloxygen, even if a $1-2 puff worked to preserve $30 of paint for the next time.
It was argued on the interweb that "any heavier than air gas will work", so I tried an experiment using 1, 1-difluroethane (aka HFC-152a) an allegedly ozone-friendly refrigerant sold inexpensively as an "air" duster. It's about twice the density of nitrogen or oxygen, so should work nicely, right?
IMG_1897.jpg
Not so fast. I had a can of polyurethane, about 80% full. Used it for a small project, then used a short blast of 1,1-difluroethane prior to closing the can. When I came back the next day, what to my wondering eyes should appear?
IMG_1895.jpg IMG_1896.jpg
But a can, collapsed, as if a good vacuum had been applied. When I pried it open there was an inrush of air, indicating a pretty good vacuum inside. OK, what then is going on?
Hypothesis 1: the 1,1-difluroethane reverted back to liquid state in the can. (Unlikely, I think, given a boiling point of -25C)
Hypothesis 2: the 1,1-difluroethane was reactive with something in the finish, making a solid or liquid product, leaving a vacuum behind (Looking at the molecule it's hard for me to posit a reaction, but I'm a biologist, not a chemist)
Hypothesis 3: 1,1-difluroethane is very soluble in oil-based poly, and simply preferred to be in the liquid phase rather than the gas phase.
It doesn't speak to the utility of the solution (unless it's reacting, which would presumably be a Bad Thing), but this was a curious, unexpected result I thought I'd share. I'd appreciate input from the chemists as to mechanism. Vacuum packing finish would be fine for preservation (works great for wine), but I'd worry about a large, mostly empty can, collapsing to the point of leakage.
For those of you who do use "canned air" (not actually air) for preserving finishes, which product do you use?
Meanwhile the search for a cheap argon tank and regulator goes on.




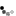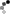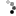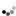
 Reply With Quote
Reply With Quote



